Difference between revisions of "Definitions, units and constants"
From Serpent Wiki
(→Definitions) |
(→Definitions) |
||
| Line 2: | Line 2: | ||
*ZA defines the element and isotope as 1000 <math>\times</math> Z + A, where Z is the proton number and A is the mass number. For example, the ZA for U-235 is 92235. | *ZA defines the element and isotope as 1000 <math>\times</math> Z + A, where Z is the proton number and A is the mass number. For example, the ZA for U-235 is 92235. | ||
| + | *ZAI defines the element, isotope and isomeric state as 10000 <math>\times</math> Z + 10 <math>\times</math> A + I, where Z is the proton number, A is the mass number and I is the isomeric state. For example, the ZAI for U-235 is 922350 and the ZAI for Am-242m is 952421. | ||
== Units == | == Units == | ||
Revision as of 00:23, 23 February 2016
Definitions
- ZA defines the element and isotope as 1000
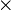 Z + A, where Z is the proton number and A is the mass number. For example, the ZA for U-235 is 92235.
Z + A, where Z is the proton number and A is the mass number. For example, the ZA for U-235 is 92235. - ZAI defines the element, isotope and isomeric state as 10000
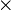 Z + 10
Z + 10 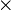 A + I, where Z is the proton number, A is the mass number and I is the isomeric state. For example, the ZAI for U-235 is 922350 and the ZAI for Am-242m is 952421.
A + I, where Z is the proton number, A is the mass number and I is the isomeric state. For example, the ZAI for U-235 is 922350 and the ZAI for Am-242m is 952421.
Units
| Quantity | Unit | Notes |
|---|---|---|
| Distance | cm | |
| Area | cm2 | |
| Volume | cm3 | |
| Time | s | depends on the case |
| Energy | MeV | |
| Microscopic cross section | b | barn = 10-24 cm2 |
| Macroscopic cross section | 1/cm | |
| Mass | g | |
| Mass density | g/cm3 | |
| Atomic density | 1024/cm3 | ( = b-1cm-1) |
| Power | W | |
| Power density | kW/g | |
| Particle flux | 1/cm2s | |
| Reaction rate | 1/cm3s | reaction rate density |
| Burnup | MWd/kgU | per total initial heavy metal |
| Burn time | days |
Constants
| Constant | Value | Units | Notes |
|---|---|---|---|
| INFTY | 1037 | N/A | Used to denote infinity |
| ZERO | 10-37 | N/A | The smallest nonzero absolute value |